Transmedics TD Cowen Conference
When I thought they couldn't do much better
TransMedics was present at the TD Cowen conference this week and shared some interesting news about the Kidney OCS, the next-generation OCS, guidance and future margins.
We are only a few days past their excellent Q4-25 but there are some information which weren’t shared then, so I thought this write up could interest some of you shareholders.
The Kidney Opportunity
This isn’t news: the kidney market is very large and has flaws that can be addressed by the OCS and its warm preservation and perfusion systems. Just like other transplanted organs, kidneys suffer from high organ loss and post-transplant complications, which have a financial impact on healthcare systems.
Today, kidney transplant post-transplant complication rate that requires the patient to go back on dialysis, that costs CMS up to $50,000-$60,000 per occurrence, called delayed graft function, is at all-time high at an incident of approximately 55%-60%.
Another nuance in kidney that’s very important to mention is CMS is financially responsible for all end-stage renal disease conditions in the United States.
The second is another very, very alarming rate, which is kidney discards or utilization of kidney donors. In the United States today, kidney utilization is at all-time low of approximately 60%.
This is the classic value added by the OCS across all organs: increasing both volume & outcomes, and it has already done wonders in livers, with concrete data proving it.
We are talking about a ~$3B market FY25 expecting to grow ~6% CAGR, maybe even more if TransMedics were to replicate its success with liver expanding the market and its market shares, which was ~36% FY25. This is napkin math but a 35% market share on a $3B market is ~$1B revenue.
Again, napkins math as we don’t know pricings for the service, etc… But it’s enough to illustrate the opportunity ahead.
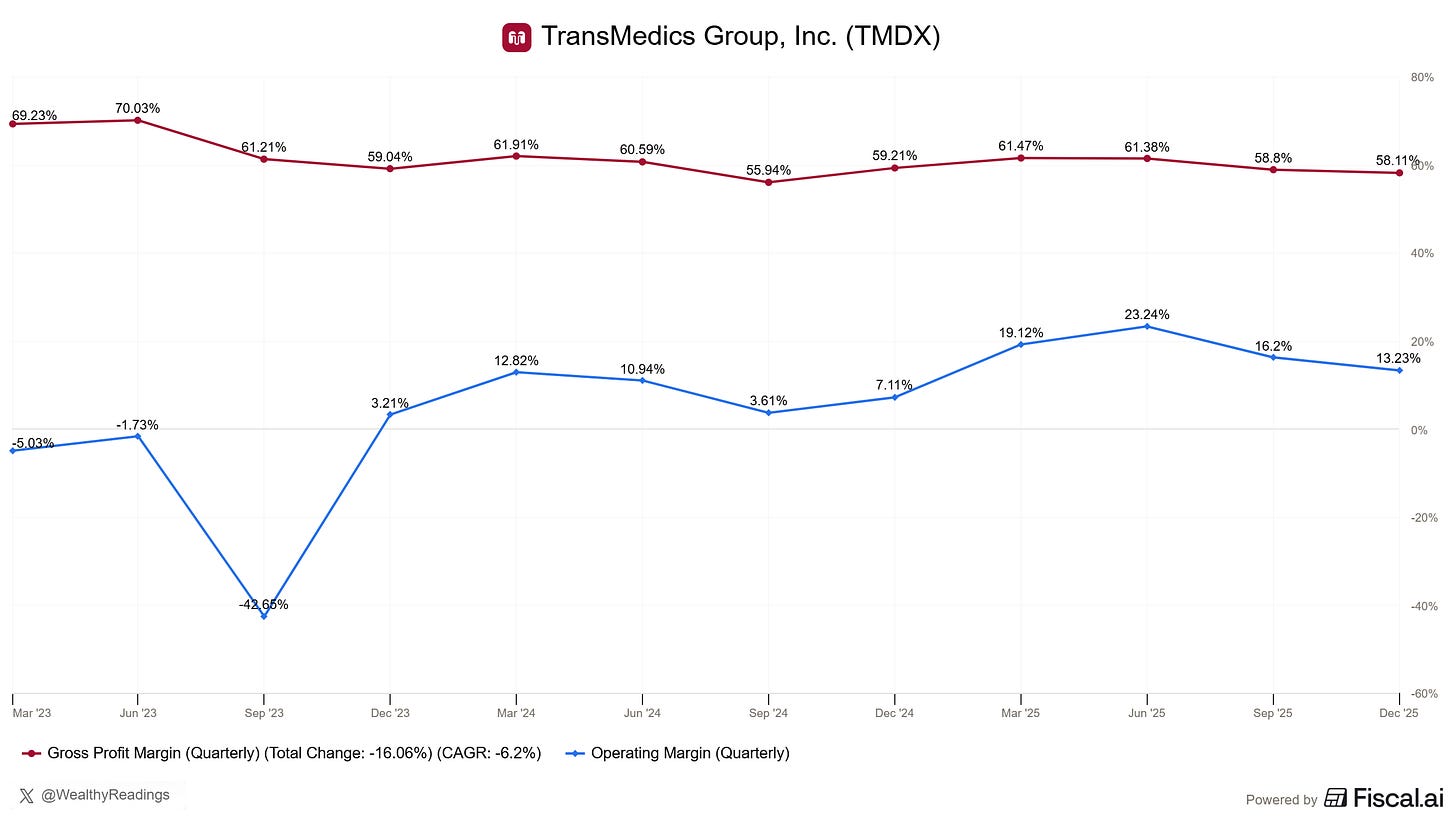
Margin Profile & Cash Generation
TransMedics is healthy in terms of cash generation but I believe the market wasn’t happy with the potential flat or shrinking margins FY26 as the company invests in European expansion, kidney OCS, Gen-3 OCS while growing expenses to run NOP services - which are a large source of growth.
As I said in my quarterly review, these short-term expenses will increase margins and cash generation in the years to come. TransMedics’ CFO explained how margins will expand in the case of the kidney Gen-3 OCS and all Gen-3 OCS platforms after that one is approved and commercialized.
There are two areas that we are really looking for to capture additional margin.
One is the device itself. The device itself will be smaller. We have less part counts, which basically will allow us to reduce the cost of assembling.
Two, right now, a lot of processes are manual. We’re expecting to do a lot of that process with robots. Today we need a clinical specialist that needs to be babysitting the organ from the moment that is placed into the device to the moment that is removed from the device for transplantation to assess the organ. Well, imagine a scenario in which with that next generation, we can do that testing, that assessment and treatment remotely via a clinical command center in our headquarters. That’s actually the vision
The kidney OCS is expected to be commercialized - at least in trials, by H1-27, not only in the U.S. but internationally.
We already presented the design to the clinical community at the American Society of Transplant Surgeons winter meeting in January. We have another milestone in June here in Boston at the American Transplant Congress, where we are aiming to have functioning devices by then. Then it’s a race through getting all the testing required to present the dossier to FDA, and hopefully by first half of next year, we’re in the clinic.
We are seriously considering having a parallel path, both releasing the product internationally and at the U.S. at the same time for clinical programs.
TransMedics is moving very fast, and the expenses of FY26 could start yielding results as soon as 2027 with volume growth from Europe and margin stabilization with the Gen-3 OCS. We’re talking about a year of spending before concrete rewards in cash generation - if only stabilization.
Nothing says everything will work perfectly. But the potential is here & is very bullish. The market will have answers quickly: one or two years is a very rapid timeframe.
Governmental Push
I shared in a previous write-up that a one bull case would be a governmental push. Today, the transplant “rulebook” in the U.S. focuses on the initial quality of the organ. An organ which doesn’t have 100% chance of yielding a perfect transplant is thrown. This creates a situation where viable organs are wasted because Organ Procurement Organizations (OPOs) would rather play it safe than risk lowering their performance scores - and therefore waste viable organs.
But the U.S. system is trying to shift toward a model that prioritizes both volume and quality, pressuring OPOs to use all viable organs and maintain high clinical standards. It is not a “transplant everything” but a move to maximize organ usage.
This would force OPOs to do everything to ensure transplants, hence adopting the best hardware and service to preserve & monitor organs from donor to recipient.
All these initiatives are mandated on the entire community of organ transplant by bipartisan effort from U.S. government, HRSA, CMS, and others. That is ongoing, and there is new language coming out of CMS, new initiative, new language, talking about potentially opening up the opportunities for new entities to become Organ Procurement Organization to maximize competitiveness and maximize transparency.
We’re the only company that has the only ischemia-free technology for organ preservation. We’re the only company that invested in vertically integrated logistics for organ transplant, 100% dedicated to organ transplant. We’re the only company that developed a very sophisticated digital ecosystem to maximize transparency, and visibility of every organ that we carry.
We will express our interest to try to play a bigger role in collaboration with existing structures in that new Modernization Act of organ transplantation in the United States.
Europe is different. Hardware is selected and purchased by national health services, governmental agencies based on governmental budgets/clinical mandates, not on a scoring system or private economics. TransMedics’ expansion in Europe depends on these governmental decisions and will grow volume by proving superior outcomes compared to traditional methods; results already demonstrated in the U.S.
They are already operating in Europe, Italy mainly, and have a healthy growing volume while management is planning to build its NOP H1-26. They aren’t spending without visibility on demand for their OCS, this expansion is the sign that European countries want OCS/NOP, that demand is here.
They know they’ll be well received in the region, they wouldn’t move otherwise.
Hearts, Lungs & Guidance
Regarding the trials, nothing much. Management shared that the onboarding rhythm will be the key indicator of how well the trials are progressing until official results are available. This is what we will need to monitor: growth in heart and lung procedures. Any acceleration means that their new hardware is a net improvement.
Now it’s showtime. Now we need to transition that momentum into enrollment, into completion of the trial, because that’s really, that’s when we know that, you know, people vote with their commitment in enrollment and adoption. We can’t wait to see that in action in Q3 and Q4.
They also communicated that even though competitors did not want to take part in the comparison arm of the trials, they’d find a way either to force them or make the comparison themselves - to my understanding. They plan to reveal in April how they will proceed if competitors continue to refuse participation.
I think the competitor spoke loudly when they were unsure of their performance against the new OCS. I think that reaction alone is very telling. We’re not gonna allow that behavior to derail or delay the trial.
We have a plan. We’re gonna execute it, and we’re gonna announce it publicly at the upcoming ISHLT in April.
Lastly, regarding those trials, management confirmed that their FY26 guidance does not include the entire trial volume. It is effectively sandbagged to give themselves room for enrollment speed.
It doesn’t include any incremental impact. We have 300 patients in the case of lung. The first 100, which more or less what we do today, that’s included. On the heart side, part A, 175, that’s current. We’re assuming that that is part of our current baseline. However, in part B, 320 hearts, we don’t have that.
We don’t know how fast we’re gonna be able to enroll because of what we have mentioned before. That one, it can be an upside, absolutely.
Not all future volume will be part of the trials. But management confirmed they intend to complete them within maximum 18 months - by end of H1-27, meaning ~800 OCS cases both organs combined, and only guided for ~275 FY26.
Question’s how fast enrollment will happen and that is a fair question. But assuming the trials proceed without issues like clear signs that the OCS is harmful, which is hard to imagine but could happen, it would be surprising if TransMedics were to only do ~275 OCS case on the ~800 expected during 75% of the trials timeframe.
A fair expectation would be to complete at least half of them - hence ~250 hearts and ~150 lungs, if not more. For context, TransMedics performed 854 heart and 88 lung cases FY25. Lungs is a drag but as this new OCS is the only technology interesting to try for the organ, many OPOs could like to take part of the trial which is better than nothing - that is speculation though.
Based on the latest quarter, ~250 heart OCS would be worth ~$36M & 100 lung OCS ~$17M, which means a ~$11M additive revenue on top of today’s guidance. Any net increase in volume translates into net increase in FY26 revenue, and that is excluding additional sandbagging related to seasonality and European expansion.
More napkins math here as the trials aren’t billed at 100% and the comparison branch of the trial will add costs to TransMedics but this remains interesting to be aware of.
I know I may look very bullish. But this is what the data suggests, and as long as those trials continue to enroll, it means they are also producing results. I see no reason not to be enthusiastic about the future as long as the data is bullish. And it is.
I think it’s very important to note that my position is not just a hypothesis or the thesis or a dream. My position is based on the fact that we are sitting here with nearly 10,000 liver transplants in the U.S. We know what the data says. The data is unequivocal in liver transplantation. The OCS is superior to cold storage. The OCS is superior to competitive technologies. The OCS is superior to back-to-base model. The OCS is superior to other perfusion technologies that try to compete with the OCS. Period. Full stop. That’s what the market is showing. That’s how the adoption curves are being, you know, printed every quarter. It’s because of that fact. Because of that, I have a high degree of confidence that, one, our superb technology, supported by a world-class clinical leaders in the field, will continue to drive adoption in liver, and liver will continue to be an engine of growth for TransMedics.
We have a long way to go. We have a long way to go because when you’re sitting with superior technology at every metric, at every level, you can evaluate it.
It will be malpractice not to use OCS.
European Expansion
A few words on expansion, which is still ongoing with more to come.
We hope that Italy will be fully functional and you can feel the breadth of Italy by end of this year. Really, it’s a huge catalyst for 2027. We hope that by end of this year, we’ll announce who’s next in Europe, and we are working with several countries in Europe, Netherlands, Belgium, France, UK, and Switzerland are the kinda the near term opportunity in addition to others, of course.
Conclusion.
I was already bullish, and I am even more so now. Not because my bias has grown, but because the data continues to support the thesis I wrote more than a year ago.
There are many execution risks for the company: expansion, the Gen-3 OCS, the trials and the kidney hardware. Many things could go wrong - no doubt about that, but at today’s price, I’d remain bullish even with average execution. I believe valuation offers a healthy risk-reward still today. Unless we see delays, cancellations, or disappointing results in one of these areas, I expect strong performances.
I will continue to monitor heart and lung trial enrollment - the only key metric for the moment, remain attentive to European expansion and follow announcements related to kidney and the Gen-3 OCS.
Regarding the stock price, I expect Q1 to break records based on recent flight data, but the key metric to watch for stock appreciation will be margins. Growth is a given.
TransMedics is my largest position and my favorite risk-reward in today’s market. I am consistently satisfied with what I hear and see, and I hope that continues for a long time. Hopefully with the stock following the trend of the fundamentals.